Thomas Brusila, Managing Director at Jaakkoo-Taara, a Finnish packaging products manufacturer serving primarily the pharmaceutical and food industries, shares his perspective on where pharmaceutical packaging is heading, and why fundamentals matter most.
Reliability
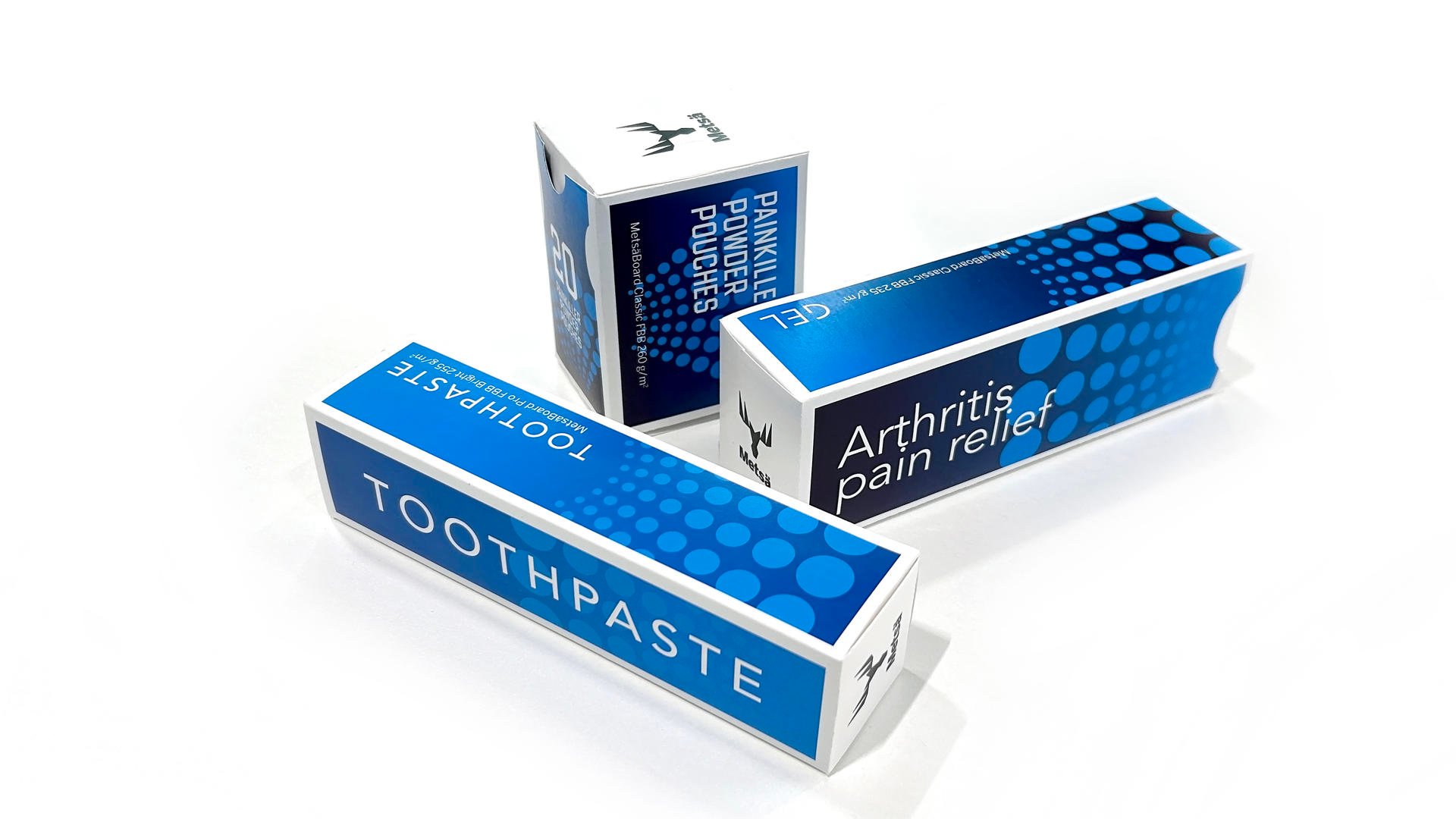
1. Safety first pharmaceutical packaging
Patient health and safety depend on packaging that functions exactly as intended, without exception.
As a result, visible anti-counterfeit and anti-tamper solutions are no longer optional – they’re industry standard. Unique serial numbers on every unit, QR codes, tear-away labels and sealed cartons are now the backbone of pharmaceutical packaging, protecting medicines and ensuring authenticity throughout the supply chain.
Behind these seemingly simple elements lies a high level of printing expertise. Depending on customer requirements, implementing these solutions can involve complex printing technologies and tightly controlled processes.
In pharmaceutical packaging, reliability isn’t just a feature, it’s a prerequisite
Quality standards
2. Rising print quality standards in pharmaceutical cartons
Print quality in pharmaceutical packaging is under closer scrutiny than ever before. Fibre-based cartons now carry increasingly refined graphics and microlevel detail, pushing precision to new limits.
Consistency is non-negotiable: colour reproduction, line sharpness and registration accuracy must remain stable from run to run, ensuring both regulatory compliance and consistent brand presentation.
At the same time, pharmaceutical manufacturers are looking to optimise the entire packaging line, ensuring smooth, reliable carton runnability and streamlined logistics.
Fresh fibre materials deliver higher strength at lower grammages, supporting faster, more efficient operations.
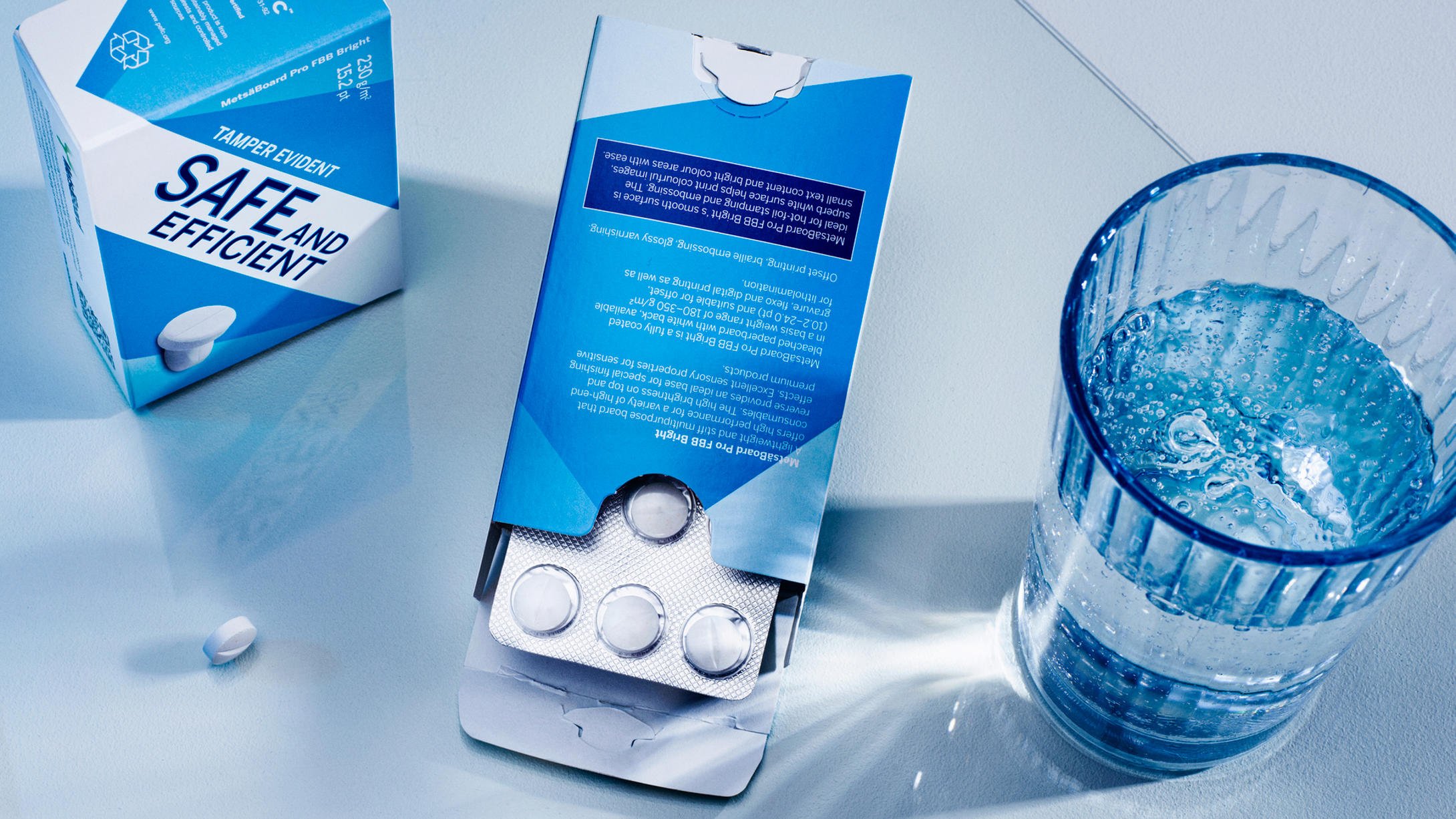
Printing technology
3. Digital printing technology in pharmaceutical packaging
Offset printing continues to dominate pharmaceutical packaging, particularly for high‑volume production. However, digital printing is steadily gaining ground as technologies mature and equipment becomes faster, more automated and more reliable.
Digital printing adds flexibility to pharmaceutical packaging production. Cost efficiencies are no longer limited to cartons alone; digital solutions are increasingly used for thin, uncoated medical leaflet papers that meet both quality expectations and regulatory requirements.
At the same time, digital workflows reduce manual complexity and help compensate for the shrinking pool of skilled offset specialists, making pharmaceutical printing more scalable.
Packaging regulations
4. How EU packaging regulations shape material choices
Evolving pharmaceutical packaging regulations are driving significant change in material selection and packaging design.
In the EU, the Packaging and Packaging Waste Regulation (PPWR) defines what types of packaging can be placed on the market and sets requirements for waste reduction, recyclability and the minimisation of empty space.
While ambitious carbon reduction targets are an important step forward, pharmaceutical packaging operates under strict safety and approval processes. These requirements can make rapid material substitution challenging, particularly when replacing plastic or aluminium components that serve critical protective functions.
Material decisions must therefore balance regulatory compliance, patient safety and environmental objectives. In pharmaceutical packaging, progress depends on making informed, compliant choices rather than pursuing change for its own sake.
Sustainability
5. Transparency and traceability as the baseline for sustainability
In pharmaceutical packaging, sustainability is defined by evidence rather than intention.
Customers now expect verified environmental and social responsibility credentials, while regulators require full traceability of raw materials throughout the value chain.
Transparency has become a baseline requirement. Packaging manufacturers must be able to demonstrate where materials originate, how they are processed and how compliance is ensured at every step.
Nordic carton manufacturers are especially well positioned to turn sustainably managed forests into a true competitive advantage and a brand asset. When sustainability is measurable, verifiable and aligned with regulatory requirements, it becomes a competitive advantage and a credible part of the brand story.